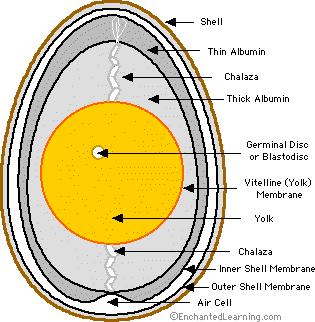
Apple Cider Vinegar also contains malic acid which acts in a similar way to lower pH levels in the gut. This stimulates appetite and improves the digestive process. Potassium assists in helping maximum nutrition to be obtained from the feed – particularly the absorption of calcium which is very important for growing chicks and laying hens.
Organisms in the gut can affect the health of chickens and compete with them for nutrition. By lowering the pH in the gut beyond that in which a specific organism can survive, the organism is eradicated. This has been demonstrated to be effective with control of pathogens such as Salmonella*, coccidiosis oocysts and internal parasites.
Apple Cider Vinegar is best given as an additive to drinking water. It can be fed to any age of bird. There are several methods of adding the vinegar to the water, they are all effective and can be adapted to suit your own requirements or regime.
The most effective sort of Apple Cider Vinegar to use is cold pressed and unfiltered as it retains more of its essential and effective vitamins and minerals.
Cautions: Do not use metal water dishes, except stainless steel – plastic is preferred. Do not use on birds which you already suspect of having excessive internal inflammation or irritation.
Two methods of giving Apple Cider Vinegar are described here. They are both effective but it is probably best to stick to one method once you decide which to follow.
Method 1 – 25ml of Apple Cider to be added to 1 litre of drinking water which is given to the hens for a period of 10-14 days to adjust pH and establish healthy gut flora. Followed by maintenance periods of either 2 days a week (the weekend for example) or one week per month. (That is a ratio of 40:1)
Method 2 – 1 ml per litre of water given on a constant basis – this is very effective for rearing chicks and growers to 8 weeks or so.