Artificial Insemination of Poultry
John L. Skinner and Louis C. Arrington
from http://www.browneggblueegg.com/Article/ArtificialInseminationOfPoultry/ArtificialInseminationOfPoultry.html
North Central Regional Extension Publication #216
Sponsored by the Extension Services of Illinois, Indiana, Iowa, Kansas, Missouri, North Dakota, Ohio, and Wisconsin.
This publication is offered as a guide for poultry hobbyists, aviculturists and others who breed ornamental fowl and want to use techniques commercial poultry breeders, particularly those in the turkey industry, have perfected to improve fertility.
When to Use Artificial Insemination
Chicken breeders may be disappointed when their better birds fail to reproduce. The birds may not mate because of shyness, physical limitations, lack of interest or social incompatibility. Unsatisfactory nutrition, age of breeders, management conditions, egg collection and holding practices, and incubation procedures can also influence production.
If birds do not reproduce when other conditions are adequate, artificial insemination may be the answer. It is relatively simple and can be used for many kinds of birds, but it requires practice and the proper equipment. It cannot, however, overcome poor management practices, poor health, genetic lethals or differences; nor will it halt early embryonic deaths.
Artificial insemination is more an art than a science. The procedure is not highly technical, but basic knowledge and appreciation of the bird’s anatomy is necessary. Success depends largely on the patience and skill of the inseminator. Wild-bird and waterfowl breeders should practice first with some common poultry type; Cornish bantams would be an excellent choices.
The Male
For best results, the male used for artificial insemination:
1. Must be mature, healthy and physically normal
2. Must be sexually active. This is especially important in birds that have a limited season. Light stimulation may be used to control the season in some varieties.
3. Must be tame, or at least not terrified when restrained or handled.
4. Should be free from external parasites. Some parasites irritate the vent area, making male organ exposure difficult and painful to the bird.
5. Should be kept apart from, but preferably in sight of, females.
6. Should not be subjected to extreme temperatures or allowed to become overheated.
The Female
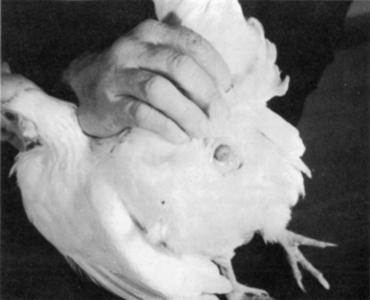
For best results, the female used for artificial insemination:
1. Must be in production, or she may be injured.
2. Must not have a hard-shelled egg in the lower part of her oviduct, so the sperm can move easily to the area where it unites with the ova.
Points to Remember
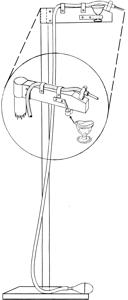
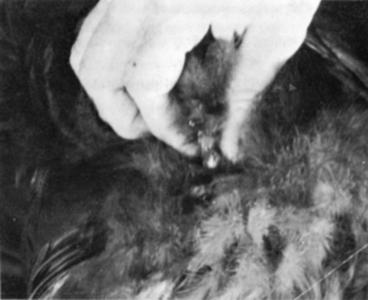
• Stimulate males and collect semen immediately after catching. Holding a male, even a tame one, for only a few minutes may interfere with collection.
• Successful semen collection usually results from an experienced operator and an experienced subject.
• First attempts at “working” inexperienced males often produce unsatisfactory results. Some males pass feces or urates as they discharge semen. Try to collect only semen; contaminated semen usually produces poor results. Withhold water and feed four to six hours before collection to lessen chances for contamination.
• The volume of semen discharged varies from bird to bird. Most males produce between 0.1 cc and 0.44 cc during each successful collection.
• Individual males vary considerably in time needed to replenish their semen supply. Normally, however, you can collect semen every two to four days without harming the birds.
• Use the semen as soon as possible. It can be held one or two hours without great loss in fertilizing capacity, or longer under controlled conditions. Don’t allow the semen to dehydrate and keep it below the body temperature of the male that produced it.
Points to Remember
• How often insemination is needed for satisfactory results varies somewhat among females. It may be best to inseminate more often at the onset of production, but once some eggs have been fertilized, once-a-week insemination is enough to maintain a satisfactory level.
• Fertile eggs can normally be obtained 48 to 96 hours after insemination and up to three weeks thereafter. The percentage of fertile eggs from a flock begins to drop between five and seven days and usually will be unsatisfactory beyond 10 days.
• Turkeys remain fertile longer than some other birds. Geese show considerable individual variation.
| WW |
| For single copies of this and other North Central Regional Extension Publications, write to: Publications Office, Cooperative Extension Service, in care of the University Listed on your left for your state. If you want information about ordering quantities of this or other Regional Publications, write or call the coordinating office for the NCR Educational Materials Project B-10 Curtiss Hall Iowa State University Ames, Iowa 50011 515-294-8802.Programs and activities of the Cooperative Extension Service are available to all potential clientele without regard to race, color, sex, national origin, or handicap. |
| W |
| Sponsored by the Extension services of Illinois, Indiana, Iowa, Kansas, Missouri, North Dakota, Ohio, and Wisconsin, in cooperation with ES-USDA.This publication is available from your Wisconsin county Extension office or from: Agricultural Bulletin Building 1535 Observatory Drive Madison, Wisconsin 53706 608-262-3346 |
| WW |
| In Cooperation with NCR Educational Materials ProjectIssued in furtherance of Cooperative Extension work, Acts of Congress of May 8 and June 30, 1914, in cooperation with the U.S. Department of Agriculture and Cooperative Extension Services of Illinois, Indiana, Iowa, Kansas, Michigan, Minnesota, Missouri, Nebraska, North Dakota, Ohio, and South Dakota. Charles F. Koval, Director, Cooperative Extension Service, University of Wisconsin, Madison, Wisconsin 5370 |
| WW |
| John l. Skinner is Professor, Department of Poultry Science, College of Agricultural and Life Sciences, University of Wisconsin-Madison and poultry and small-animal specialist, Cooperative Extension Service, University of Wisconsin-Extension.Louis C. Arrington is professor, Department of Poultry Science, College of Agricultural and Life Sciences, University of Wisconsin-Madison and poultry specialist, Cooperative Extension Service, University of Wisconsin-Extension. |
North Central region Publication Extension No. 216